Atomic mass of al4/9/2023  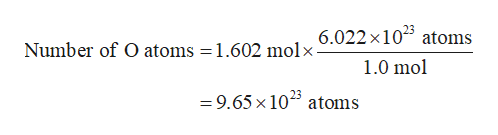
When an organism dies, it stops taking in carbon-14, so the ratio of carbon-14 to carbon-12 in its remains, such as fossilized bones, will decline as carbon-14 decays gradually to nitrogen-14 2 ^2 2 squared. Mass of aluminium, w8.1 g Number of atoms of Al,N Al Molar massMass ×N A Mw×N A 278.1×N A There are 4 atoms present per unit cell as it crystallizes in a face-centred cubic (f.c.c.) structure. 
Multiplying the number of atoms of each of these elements by its respective atomic weight and then adding them, the molecular weight of Al2O3 is 101.961276 grams per mole. As animals eat the plants, or eat other animals that ate plants, the concentrations of carbon-14 in their bodies will also match the atmospheric concentration. Atomic Mass Average: 26.98154 Boiling Point: 2740K 2467C 4473F Coefficient of lineal thermal expansion/K-1: 23.03E Conductivity. Using the periodic table, a student can find the atomic masses of aluminum as 26.9815386 grams per mole and oxygen as 16.9994 grams per mole. As plants pull carbon dioxide from the air to make sugars, the relative amount of carbon-14 in their tissues will be equal to the concentration of carbon-14 in the atmosphere. Atomic mass of Al 26.982 g/mol, Atomic mass of O 15.999 g/mol, Atomic mass of H 1.00794 g/mol Molar mass of Al (OH) 3 1 atomic mass of Al + 3 atomic mass of O + 3 atomic mass of H 1 26.98g/mol + 3 15.999 g/mol + 3 1.00794 g/mol 26.982 g/mol + 47.997 g/mol + 3.0238 g/mol 78. Now, 27 g of aluminium has ions 6.022 × 1023 aluminium ions. 
This means that 1 mole of aluminium atoms ( or aluminium ions ) has mass of 27 grams, and it contains 6.022 × 1023 aluminium ions. These forms of carbon are found in the atmosphere in relatively constant proportions, with carbon-12 as the major form at about 99%, carbon-13 as a minor form at about 1%, and carbon-14 present only in tiny amounts 1 ^1 1 start superscript, 1, end superscript. The atomic mass of aluminium is given to be 27 u. For example, carbon is normally present in the atmosphere in the form of gases like carbon dioxide, and it exists in three isotopic forms: carbon-12 and carbon-13, which are stable, and carbon-14, which is radioactive. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
